|
3/30/2023 0 Comments Definition of ion bonding 
Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their electrons to achieve a stable electron configuration. As a result, weakly electronegative atoms tend to distort their electron cloud and form cations. Atoms that are weakly electronegative (such as alkali metals) have relatively few valence electrons, which can easily be shared with atoms that are strongly electronegative. Atoms that are strongly electronegative (as is the case with halogens) often have only one or two empty orbitals in their valence shell, and frequently bond with other molecules or gain electrons to form anions. OverviewĪtoms that have an almost full or almost empty valence shell tend to be very reactive. They also tend to be soluble in water the stronger the cohesive forces, the lower the solubility. The higher the charges the stronger the cohesive forces and the higher the melting point. Ionic compounds generally have a high melting point, depending on the charge of the ions they consist of. Ionic compounds conduct electricity when molten or in solution, typically not when solid. Bonds with partially ionic and partially covalent character are called polar covalent bonds. Thus, the term "ionic bonding" is given when the ionic character is greater than the covalent character – that is, a bond in which there is a large difference in electronegativity between the two atoms, causing the bonding to be more polar (ionic) than in covalent bonding where electrons are shared more equally. It is important to recognize that clean ionic bonding - in which one atom or molecule completely transfers an electron to another - cannot exist: all ionic compounds have some degree of covalent bonding, or electron sharing. In simpler words, an ionic bond results from the transfer of electrons from a metal to a non-metal in order to obtain a full valence shell for both atoms. In the simplest case, the cation is a metal atom and the anion is a nonmetal atom, but these ions can be of a more complex nature, e.g. This transfer of electrons is known as electrovalence in contrast to covalence. Atoms that lose electrons make positively charged ions (called cations). Atoms that gain electrons make negatively charged ions (called anions). Ions are atoms (or groups of atoms) with an electrostatic charge. It is one of the main types of bonding, along with covalent bonding and metallic bonding. 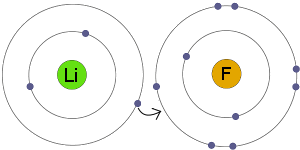
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. The oppositely charged ions – typically a great many of them – are then attracted to each other to form solid sodium fluoride. Sodium loses its outer electron to give it a stable electron configuration, and this electron enters the fluorine atom exothermically. Sodium and fluorine atoms undergoing a redox reaction to form sodium ions and fluoride ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
